In other words, the number of valence electrons for a transition metal is equal to how many spaces across the periodic table it is. The valence electrons for transition metals are equal to the number of s-electrons plus the number of d-electrons. For non-transition metals, we count to 8, but for transition metals, we count to 12. Looking at the orbitals explains how valence electrons work for transition metals. For example, oxygen has 6 valence electrons, these six electrons fill up the 2s orbital, and partially fill the 2p subshells (2s 22p 4). The shells after (ignoring transition metals) represent the s and p-orbitals. Using the variable n to represent the number of the valence electron shell, write the valence shell electron configuration for each group. So what does this have to do with our shells? The first "shell" represents the 1s orbital. The shape of the periodic table reflects the order in which electron shells and subshells fill with electrons. The actual layout of the periodic table is based on the grouping of the elements according to chemical properties. For example, carbon has an electron configuration of 1s 22s 22p 2. The reason for this choice will become more apparent in Chapter 3 when we discuss valence and electron configuration in more detail. The way we count our electrons is by moving from right to left, starting at the beginning of the table. Valence electrons: The electrons present in the outermost orbit of an atom are called valence electrons. F-orbitals start appearing in the lanthanides and actinides (the separated two rows).

P-orbitals start appearing in period 2, and d-orbitals start appearing in period 4 (though they start counting at 3).

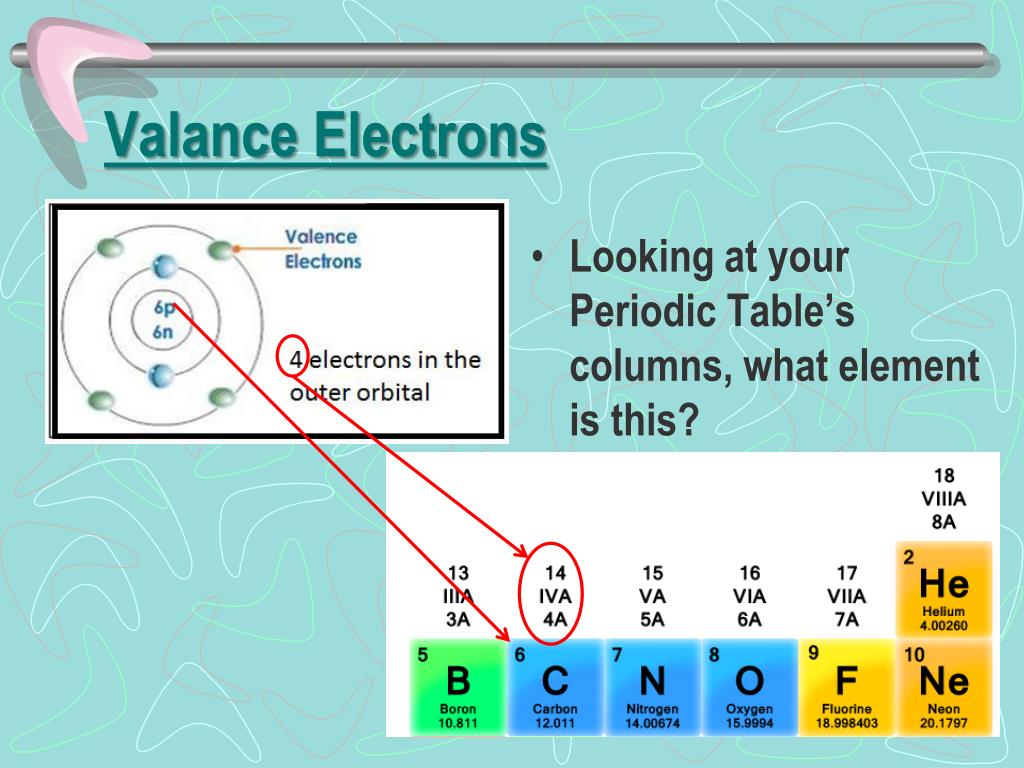
Valence electrons are typically the electrons that remain after all of an atom’s inner subshells have been filled with protons. In the second period elements, the two electrons in the 1s 1 s sublevel are called inner-shell electrons and are not involved directly in the elements reactivity, or in the formation of compounds. Finding Valence Electrons for Transition Metals. Valence electrons are the electrons in the highest occupied principal energy level of an atom. It has gained two electrons, its swiped it from somebody else. Due to the fact that oxygen is found in group 16 of the periodic table, it contains six valence electrons. This is the reason why H is always a terminal atom and never a central atom. So these elements also like to attract electrons. Hydrogen only needs to form one bond to complete a duet of electrons. Atom (Group number)īecause hydrogen only needs two electrons to fill its valence shell, it follows the duet rule. Table showing 4 different atoms, each of their number of bonds, and each of their number of lone pairs.
In each case, the sum of the number of bonds and the number of lone pairs is 4, which is equivalent to eight (octet) electrons. The number of electrons required to obtain an octet determines the number of covalent bonds an atom can form. Oxygen and other atoms in group 16 obtain an octet by forming two covalent bonds: To obtain an octet, these atoms form three covalent bonds, as in NH 3 (ammonia). Group 15 elements such as nitrogen have five valence electrons in the atomic Lewis symbol: one lone pair and three unpaired electrons. The transition elements and inner transition elements also do not follow the octet rule since they have d and f electrons involved in their valence shells. The paper introduces Chemdex, a freely accessible web-based database of over 70 000 compounds characterised by crystallography from across the periodic table. Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule and only needs to form one bond. These four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in CCl 4 (carbon tetrachloride) and silicon in SiH 4 (silane). For example, each atom of a group 14 element has four electrons in its outermost shell and therefore requires four more electrons to reach an octet. The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). From left to right: water molecule, ammonia molecule, and methane molecule
